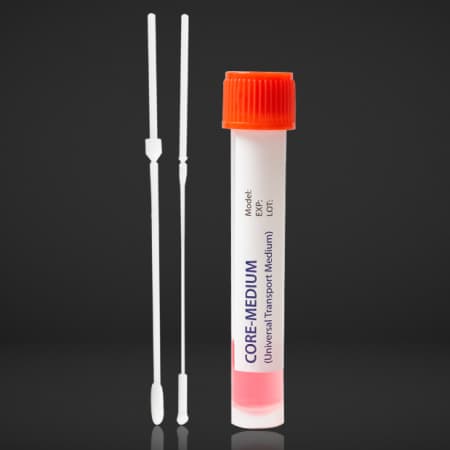
CORE-MEDIUM System
Negotiable Min Order Quantity Unit
- Required Quantity
-
- Place of Origin
- South Korea
- Payment Terms
- Negotiable
- Production method
- Negotiable
- Shipping / Lead Time
- Negotiable / Negotiable
- Keyword
- diagnostic devices
- Category
- Monitoring & Diagnostic Equipment
Apply a video call to the Supplier

Incore Co.,Ltd.
- Country / Year Established
-
 South Korea
/
2014
South Korea
/
2014
- Business type
- Manufacturer
- Verified Certificate
-
5


| Product name | CORE-MEDIUM System | Certification | - |
|---|---|---|---|
| Category | Monitoring & Diagnostic Equipment | Ingredients | - |
| Keyword | diagnostic devices | Unit Size | - |
| Brand name | - | Unit Weigh | - |
| origin | South Korea | Stock | - |
| Supply type | - | HS code | - |
Product Information
The Core Medium System is intended nfor the collection and transport of clinical specimens containing viruses, chlamydia, mycoplasma or ureapolasma, etc. from the collection site to the testing laboratory.
| No | Model | Length | Break Point | Part Used(Product Composition) |
| 1 | IC-NS01 | 140mm | 90mm | Nasal Cavity |
| 2 | IC-OS01 | 140mm | 90mm | Oral Cavity |
| 3 | IC-ND01 | 140mm | 90mm | Nasal Cavity(IC-NS01 x 2) |
| 4 | IC-ON01 | 140mm | 90mm | Nasal Cavity & Oral Cavity(IC-NS01 + IC-OS01) |
| 5 | IC-ON02 | 140mm | 90mm | Nasal Cavity & Oral Cavity(IC-NS01 x 2 + IC-OS01) |
| 6 | IC-OD01 | 140mm | 90mm | Oral Cavity(IC-OS01 x 2) |
 | No | Model | Measure of Capacity(mL) | Color of Cap |
| 1 | IC-UTM01W | 1mL | White | |
| 2 | IC-UTM02W | 2mL | White | |
| 3 | IC-UTM03W | 3mL | White | |
| 4 | IC-UTM01R | 1mL | Red | |
| 5 | IC-UTM02R | 2mL | Red | |
| 6 | IC-UTM03R | 3mL | Red |
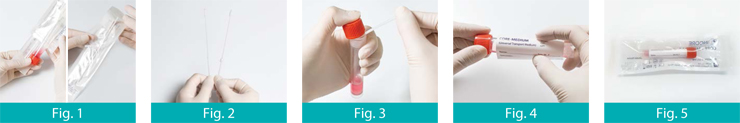
1. Open the Zipper bag
2. Take the products out of its bag carefully and open the peel respectively
3. Collect the specimen by swab; to prevent the risk of contamination, make sure that the swab tip comes into contact with the collection site only.
4. After collecting insert the swab into the tube. Please take care not to spill the contents of the tube.
5. Bend the swab stick to break it off at the breaking point.
6. After screwing the cap of the tube tightly, record information about the patient on the label and attach it on the tube.
7. Send to the laboratory for immediate analysis.
B2B Trade
| Price (FOB) | Negotiable | transportation | - |
|---|---|---|---|
| MOQ | Negotiable | Leadtime | Negotiable |
| Payment Options | Negotiable | Shipping time | Negotiable |

- President
- KIM DONG TAK
- Address
- Hyeoksin daero 78gil 11, Dong-gu, Daegu, Korea
- Product Category
- Wound Dressing Equipments
- Year Established
- 2014
- Company introduction
-
INCORE CO., LTD offers safe disposable medical devices (Core-Forcep, Core- Clip, Core-Knife, Core-Snare, Core-Injector, Core-HotBiopsy) with no risk of infection, and hemostatic gauze ,HEMOBLOCK for topical hemostasis. We produce a variety of medical devices and medical supplies. Beyond the manufacture of medical devices, we research and develop products that reflect the requirements of medical specialists. Differentiated competitiveness based on R&D leads the development of endoscopic medical devices. We have grown into a company with a promising future. In addition, in order to globalize, exports started in Vietnam in 2018 and we are exporting to Southeast Asia, including Vietnam, Thailand, and Myanmar, in earnest in 2019. We are expecting that we can export to Russia around 2020 as we proceed with Russian licensing and branching.
- Main Markets
-
 Austria
Austria
 Croatia
Croatia
 Iran
Iran
 Myanmar
Myanmar
 Russia
Russia
 South Korea
South Korea
 Thailand
Thailand
 Viet Nam
Viet Nam
- Main Product
Related Products

Urine Analysis Test Strip Self-Stik Series

URIVET10

Best Companion
Osteopro (X-ray Bone densitometer, DEXA)
Contour Test strips, Accu chek, One Touch, On Call Plus